- Molybdenum and its applications
- Molybdenum and its applications
- Role of molybdenum
- Mining & processing
- Iron, steel and other alloys
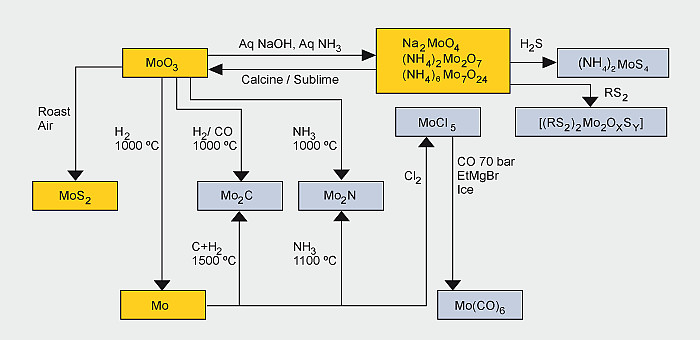
- Molybdenum metal, alloys and chemicals
- Applications by industry
- Molybdenum for life
- Health, Safety & Environment
- Health, Safety & Environment
- Introduction
- Turkiye-REACH Letter of Access Applications
- REACH MoConsortium (MoCon)
- IMOA Assaying Guideline Procedures
- OECD Mutual Acceptance of Data (MAD)
- IMOA HSE research publications
- Database of Molybdenum in Human Health and the Environment
- K-REACH
- IMOA Life Cycle Assessment Program
- Globally Harmonised System (GHS)
- Resource Center
- Resource Center
- Recources
- MolyReview
- Molybdenum videos
- AIA webinars
-
Conference proceedings and papers
- 2024 Vienna Molybdenum Symposium Mo4Steel
- 2018 Shanghai Steel Symposium proceedings
- 2013 Korea Symposium on Mo/Nb Alloying in Steels
- 2011 Taiwan Symposium on Mo/Nb Alloying in Steels
- 2010 International Seminar on the Applications of Mo in Steels
- 2007 International Experts Seminar on Stainless Steel in Structures
- Infographics
- Facts and figures
- Stainless Solutions e-newsletter
- Knowledge Center
- Knowledge Center
- Introduction
- Knowledge Center
- IMOA